Introduction
The African Dwarf Frog is a small animal, usually measuring 4-6cm in length (snout to vent) when fully grown, and is remarkably easy to keep as a pet. As a completely aquatic frog it is usually found on display in pet shops in the aquaria alongside and even in the tanks with fish. While carnivorous, African Dwarf Frog s will not attack fish larger than it can swallow and so can safely be placed in a populated tank (Truckenbrodt, 2011). These frogs have been used as model organisms by scientists for decades, but are increasingly kept as a pets in the both the USA and the UK today. With a little care and attention, these diminutive amphibians commonly live for 4-7 years and can be an ideal pet in any home.
Identification
Several terms, including ‘African Dwarf Frog’, ‘African Dwarf Clawed Frog’, ‘West African Clawed Toad’ and ‘Zaire Dwarf Clawed Frog’ have all been applied to describe the animal commonly kept as a pet, but the terms have often been used to refer to a number of different species and subspecies. For the most part, they have been applied to various members of the Hymenochirus genus of frogs; the most common recipient of the name is Hymenochirus boettgeri. The acronyms ADCF, ADF and DAF are commonly used as shorthand; this essay will use ADF (African Dwarf Frog) and, unless specified otherwise, will be referring to H. boettgeri as the frog of choice.
Clearly, in the pet trade in particular, it can be hard to ascertain which exact species an ADF belongs to. However, this will not pose a problem to the average pet keeper as all members of Hymenochirus will thrive if the basic habitat conditions are met.
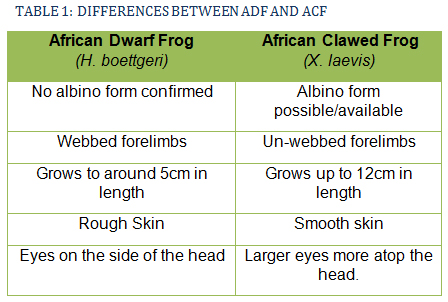
It is important to note, however, that in many pet stores the African Clawed Frog (Xenopus laevis) is kept with and sold as ADF. The African Clawed Frog is a particularly voracious predator and will commonly attack and kill fish larger than itself, ripping them apart with powerful clawed hind feet. As such, it is very important not to mistake one for the other especially if introducing into a populated fish tank. Whilst ADF can happily co-exist alongside other fish, the introduction of X. laevis into a populated tank will likely result in no fish and one well-fed frog.
Another, arguably more important reason for accurate identification is that X. laevis is a potential carrier of chytrid fungus, an infection which can be lethal to ADF.
Table 1 describes several key ways to differentiate H. boettgeri from X. laevis.
Husbandry
Tank Requirements
The ADF is a fully aquatic frog, and must be kept in a suitable tank. They will rise to gulp air from the surface every 5 minutes or so, but there is no need for the provision of any ‘dry land’ area. Any aquarium suitable for fish is also suitable for housing ADFs. An appropriate sized aquarium for a single frog should ideally be no smaller than 5 gallons. A lid is required on the tank and it is recommended that the water level is kept at least 2 inches below the lip of the tank. The gap will allow suitable gas exchange while the lid will prevent escape.
As the name suggests, these frogs are from tropical climes; they are native to central Africa. Therefore, the ultimate aim for any tank, regardless of the method of set up (planted tank or filtered) should be a maintained water temperature of 24-25°C, a water pH of around 7.6 (Rabb, 1963) and adequate recycling/removal of nitrogenous waste.
ADFs natural habitat is tropical ponds or flooded forests with light currents at most and plenty of vegetation. As such, they should be provided with at least one patch of dense vegetation to hide in, but they will happily hide in any place they can fit. ADFs will dig under plants and rocks as they forage for food, therefore any plants should be well anchored to prevent collapse. The same is true of any rocks or other tank decoration used – care should be taken that these will not collapse, possibly trapping, injuring or killing the frog.
If a filter is used it should provide only a gentle current. It is possible for the frog’s small limbs to become trapped in an intake and drown, therefore a lattice or similar covering over the intakes is recommended in order to prevent this. In a well planted tank, the plants and soil bacteria will provide the necessary recycling of nitrogenous waste.
Uneaten food, shed skin and dead plant material should be removed from the tank, on a daily basis. Water changes of 20-30% every week is recommended (in the planted tank this may be less).
ADFs feed by shovelling or sucking food into their mouths. Because of this, it is important to ensure that if gravel is used as a substrate, it must be large enough that it will not be swallowed. Swallowing gravel can quickly lead to intestinal blockage and death in the ADF. Smooth gravel is also preferred to reduce the risk of laceration of the skin. Sand is generally more favourable as a substrate as it can be easily spat back out during foraging and poses limited risk of physical injury.
Feeding
Despite lacking tongues or teeth – like all members of the pipidae family of frogs – ADFs are carnivores and are best fed on a variety of raw meat. They will eat tuna, shrimp, bloodworm, earthworm, trout, and salmon. If the food is frozen beforehand, then it will sink and break apart easier. They will also eat plankton (e.g. daphnia) some pellet foods and, when young, flaked food. If sharing a tank with fish, care should be taken to ensure that the frogs get their fair share of food – possibly by feeding simultaneously, but at different areas of the tank.
Stories of bloodworms and mealworms eating through the guts of frogs, if fed live, are so far anecdotal only, but in any case these should not be fed in large quantities as they are high in fat, low in nutrition and could lead to obesity. As with most pets, a good variety of food should keep ADF happy and healthy.
Breeding
Breeding behaviour, in particular clasping and spawning behaviours have been described by Österdahl and Olsson (1962). Mating begins when the male clasps the female just above her rear legs. This is known as amplexus and is shown in figure 2. Oviposition (laying and fertilisation of the eggs) occurs at the surface of the water. This usually occurs over the course of a single night, but can last for two nights with the pair resting during the day. Each spawning releases a dozen eggs at most, but with up to 50 ovipositions in a single spawning session, a single pair of frogs can produce between 150-200 eggs.
Those eggs which are dislodged from or do not stick at the water surface will sink to the bottom of the aquarium (or stick to plants) and likely be consumed by adults in the normal process of foraging. The eggs themselves are visible to the naked eye, averaging around 1mm (including their jelly coat).
Sokol (1962) gives an extensive account of the details in the early lifecycle of H. boettgeri. Temperature is particularly important in breeding; eggs will not hatch at temperatures below 22°C. If conditions are good, eggs will hatch within 24 hours of oviposition. Over the next 4-5 days, the larvae remain at the water surface or on submerged plants, attached by a mucous thread. After this period, free swimming, silver-grey coloured larvae will be observable. When fully grown, tadpoles measure about 21mm.
Veterinary Care
While all vets should be able to provide basic care, those specialising in amphibians can be located via the Association of Reptile and Amphibian Veterinarians at http://www.arva.org.
The importance of the skin should always be kept in mind when handling amphibians; it is one of the most important organs, playing a vital role in respiration, water absorption, nutrient balance, acid balance and immune system function. The skin secretions that aid these roles also make amphibians slippery and difficult to handle (Fowler, 2008).
Appropriate handling techniques
In general, ADF should never be removed from the water, except for care by a vet. The combination of their small size and porous skin can result in fatal dehydration within 15 minutes following removal from the water. Removal should be done using a soft net, covering the opening to ensure ADF doesn’t jump out.
ADF should never be handled with contaminated hands; tiny amounts of natural oils from human skin, soap and/or detergents could harm the animal by being absorbed through the skin. If bare hands are used, they should be wet to help avoid dehydration, but un-powdered gloves are preferred (Stoker, 2005). It is also important to use fresh gloves/wash hands thoroughly between specimens as contaminants and infection agents – such as chytrid fungus – can be transmitted via contact.
Any handling should be performed on a damp cloth to prevent the frog from drying out. If tap water is used, it should be de-chlorinated by letting it stand for 24 hours.
Given the slippery nature of the frog, gauze can be used to establish a firm grip for injections, but if suturing is required the frog should be anesthetised first – by submersion up to the nostrils or washing with anaesthetic – to prevent any movement (Fowler, 2008 and Stoker, 2008).
Health Problems
Chytridiomycosis
Initially discovered in Australian rainforest frogs in 1993, chytridiomycosis is a serious disease caused by infection of the skin by the Batrachochytrium dendrobatidis (Bd) chytrid fungus. It is usually fatal and has been responsible for massive declines in amphibian numbers worldwide (Raverty and Reynolds, 2001).
Bd can be carried by tadpoles, but only affects adult amphibians (Berger et al. 1999). It infects the upper layers of the skin (mainly the stratum cornea) where it feeds on the keratin in the cells. In amphibians, the skin responds by growing thicker – hyperplasia – which reduces its ability to act as a permeable barrier for electrolytes. This in turn leads to an imbalance in the blood chemistry which leads to heart failure (Voyles et al., 2009).
Signs of chytrid infection include lethargy, loss of appetite and thrashing at the surface of the water, but exact diagnosis requires histological examination (Berger et al. 1999; Scott and Vance, 2007). When signs do appear, however, it is often too late for effective treatment.
Treatment with Terbinafine Hydrochloride is recommended for pets. Complete sterilisation of contaminated habitats and tanks – including safe disposal of plants and soil to avoid environmental contamination – is also required.
As always, and given the serious nature of this disease, consultation with a vet professional is the best first action in any suspected case.
Red Leg
The name of this disease comes from the red lesions that sometimes form on the hind legs, but the name is misleading as many frogs die from this affliction without showing any outward signs of infection. The disease is effectively septicaemia caused by the bacterium Aeromonas hydrophila –a bacterium normally found on the frogs skin, but non-problematic until it gets into the blood, usually through injury (Berger et al., 1999). Other signs include muscular spasms, lethargy and vomiting.
Treatment using 0.6% saline solution can be effective, but prevention via good hygiene and tank maintenance is best.
Subcutaneous Emphysema
Also known as ‘balloon frogs’, due to their appearance caused by a build-up of air under the skin. No definite cause has been identified. The air can be removed via a syringe or needle and there is usually no lasting damage to the frog. This is a fairly common occurrence in captive frogs.
Potential Health Hazards
Like all amphibians, ADF is a potential source of Salmonella. Transmission to humans is via handling or contact with the tank or ADF itself. It is recommended that those most at risk (under 5s, elderly or those with compromised immune systems) do not keep any amphibians in their home (CDC, 2009). To prevent transmission, hands should always be washed thoroughly after contact. Cleaning of the tank and habitat items should never be done in sinks where food preparation occurs.
Conclusion
When buying any pet, the first decision to make is to buy from a respected, responsible trader. Doing so will help to avoid the most common sources of illness and disease. Then, with minimal effort and simple precautions, these frogs can provide quiet, safe and dignified companionship for years at a time.
References
Berger L., Speare R. and Kent A. 1999. Diagnosis of chytridiomycosis in amphibians by histologic examination. [online] Available at: < Diagnosis of chytridiomycosis in amphibians by histologic examination. > [Accessed 20th March 2013].
CDC, 2009. Investigation Update: Outbreak of Human Salmonella Typhimurium Infections Associated with Contact with Water Frogs. [online] Available at: < http://www.cdc.gov/salmonella/typh1209/index.html> [Accessed 20th March 2013]
Fowler M. E., 2008. Restraint and Handling of Wild and Domestic Animals. Danvers, MA.: Wiley-Blackwell.
Österdahl L. and Olsson R., 1963. The Sexual Behaviour of Hymenochirus boettgeri. Oikos. (14(1), p. 35-43. Available through: Edinburgh Napier University website <http://nuinlink.napier.ac.uk> [Accessed 26th February 2013].
Rabb G. B. and Rabb M. S., 1963. On the behavior and breeding biology of the African pipid frog: Hymenochirus boettgeri. Zeitschrift für Tierpsychologie. [e-journal] 20(2), p.215-241.
Sokol O. M., 1962. The Tadpole of Hymenochirus boettgeri. American Society of Ichthyologists and Herpetologists. [e-journal] 1962(2), p. 272-284. Available through: Edinburgh Napier University website <http://nuinlink.napier.ac.uk> [Accessed 26th February 2013].
Stoker L., 2005. Practical Wildlife Care. Oxford: Blackwell Publishing
Scott B. and Vance J., 2007. Diagnosis of Chytridiomycosis in Commercially Purchased African Dwarf Frogs. [online] Available at: <http://www.flippersandfins.net/Chytridiomycosis.htm> [Accessed 20th March 2013]
Raverty S. and Reynolds T., 2001. Cutaneous chytridiomycosis in dwarf aquatic frogs (Hymenochirus boettgeri) originating from southeast Asia and in a western toad (Bufo boreas) from northeastern British Columbia. Canadian Veterinary Journal. [ejournal] 42, p.385-386. Available through: Edinburgh Napier University website <http://nuinlink.napier.ac.uk> [Accessed 2nd March 2013].
Truckenbrodt M. 2011. pipidae.net [online] Available at: <www.pipidae.net> [Accessed 6th March 2013]
Voyles, J., S. Young, L. Berger, C. Campbell, W.F. Voyles, A. Dinudom, D. Cook, R. Webb, R.A. Alford, L.F. Skerratt, and Speare R., 2009. Pathogenesis of chytridiomycosis, a cause of catastrophic amphibian declines. Science 326:582-585.

Your link to the ARAV is faulty and leads to an Arkansas educational site. Here’s the correct link: http://arav.org/